Training program for Master’s Degree in Zhejiang University(Major Pharmaceutica
2010-09-10 |
Training program for Master’s Degree in Zhejiang University
College Pharmaceutical Sciences
Major Pharmaceutical analysis (Code: 100704 )
(First Level subject: Pharmaceutical Sciences )
1. Aim
To train high-level researchers, teachers and specialized personnel, with professional knowledge and advanced technologies, for drug quality inspection, clinical pharmacy, drug metabolism and pharmacokinetic research.
2. Study period 2.5 years
3. Research topics
1. Drug transport and metabolic analysis
2. Chiral biopharmaceutical analysis
3. Study and application of drug metabolic enzyme transduced cells
4. Drug metabolism and pharmacokinetics of traditional Chinese medicine
5. Whole Process control of drug manufacturing
6. Drug quality analysis
7. Discovery of novel drugs and computer assisted drug analysis
8. Proteomics and metabolomics of drugs
9. Drug manufacturing process analysis, modeling and control
10. Clinical pharmacy study
4. Coursework requirement
A minimum credits of 26,including 6 credits for public courses, 10 credits for degree courses, 8 credits for elective courses, and 2 credits for reading report.
5. Other requirements
4. Reading report:4 reading reports and at least 2 public presentation related to the research
topic are required.
5. Dissertation proposal:a dissertation proposal hearing is required in one year after
admission. A written form must be handed in the first month of the second year.
6. Papers required:refer to the degree requirements.
Course syllabus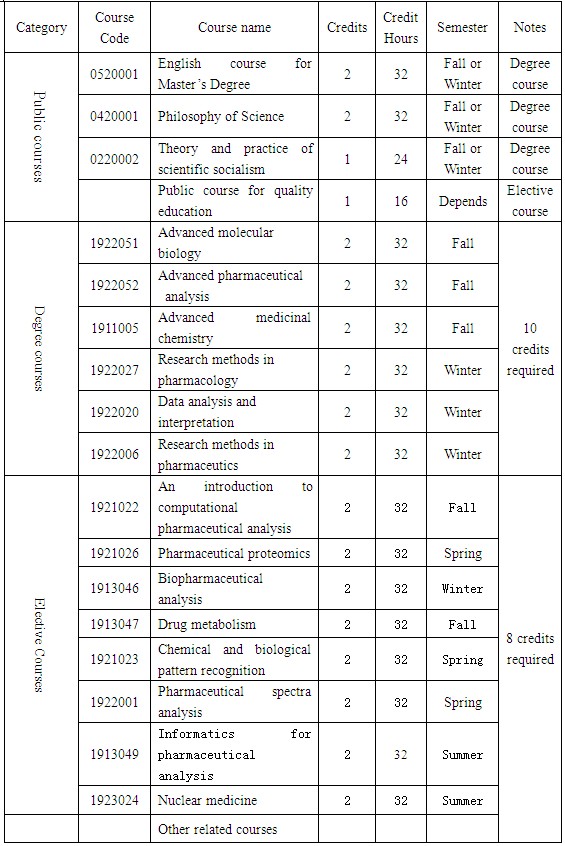
Training program for Doctor’s Degree in Zhejiang University
College Pharmaceutical Sciences
Major Pharmaceutical analysis (Code: 100704 )
(First Level subject: Pharmaceutical Sciences )
1. Aim
To train high-level researchers, teachers and specialized personnel, with professional knowledge and advanced technologies, for drug quality inspection, clinical pharmacy, drug metabolism and pharmacokinetic research.
2. Study period 3.5 years
3. Research topics
1. Drug transport and metabolic analysis
2. Chiral biopharmaceutical analysis
3. Study and application of drug metabolic enzyme transduced cells
4. Drug metabolism and pharmacokinetics of traditional Chinese medicine
5. Whole Process control of drug manufacturing
6. Drug quality analysis
7. Discovery of novel drugs and computer assisted drug analysis
8. Proteomics and metabolomics of drugs
9. Drug manufacturing process analysis, modeling and control
4. Coursework requirement
A minimum credits of 14,including 4 credits for public courses, 2 credits for special degree courses, 6 credits for elective courses, and 2 credits for reading report.
5. Other requirements
1. Reading report:6 reading reports and at least 3 public presentation related to the research
topic are required.
2. Dissertation proposal:a dissertation proposal hearing is required in two years after
admission. A written form must be handed in the first month of the third year.
3. Papers required:refer to the degree requirements.
Course syllabus
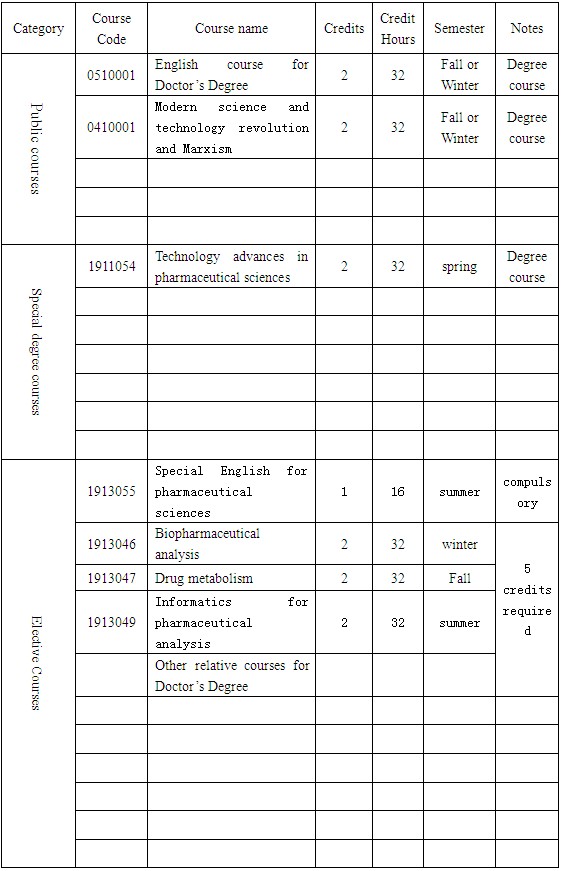

Training program for Doctor’s Degree in Zhejiang University
College Pharmaceutical Sciences
Major Pharmaceutical analysis (Code: 100704 )
(First Level subject: pharmaceutical Sciences )
1. Aim
To train high-level researchers, teachers and specialized personnel, with professional knowledge and advanced technologies, for drug quality inspection, clinical pharmacy, drug metabolism and pharmacokinetic research.
2. Study period 5 years
3. Research topics
1. Drug transport and metabolic analysis
2. Chiral biopharmaceutical analysis
3. Study and application of drug metabolic enzyme transduced cells
4. Drug metabolism and pharmacokinetics of traditional Chinese medicine
5. Whole Process control of drug manufacturing
6. Drug quality analysis
7. Discovery of novel drugs and computer assisted drug analysis
8. Proteomics and metabolomics of drugs
9. Drug manufacturing process analysis, modeling and control
10. Study of clinical pharmacy
4. Coursework requirement
A minimum credits of 38,including 8 credits for public courses, 12 credits for special degree courses, 14 credits for elective courses, and 4 credits for reading report.
5. Other requirements
1. Reading report:10 reading reports and at least 5 public presentation related to the
research topic are required.
2. Dissertation proposal:a dissertation proposal hearing is required in three years after
admission. A written form must be handed in the first month of the fourth year.
3. Papers required:refer to the degree requirements.
Course syllabus
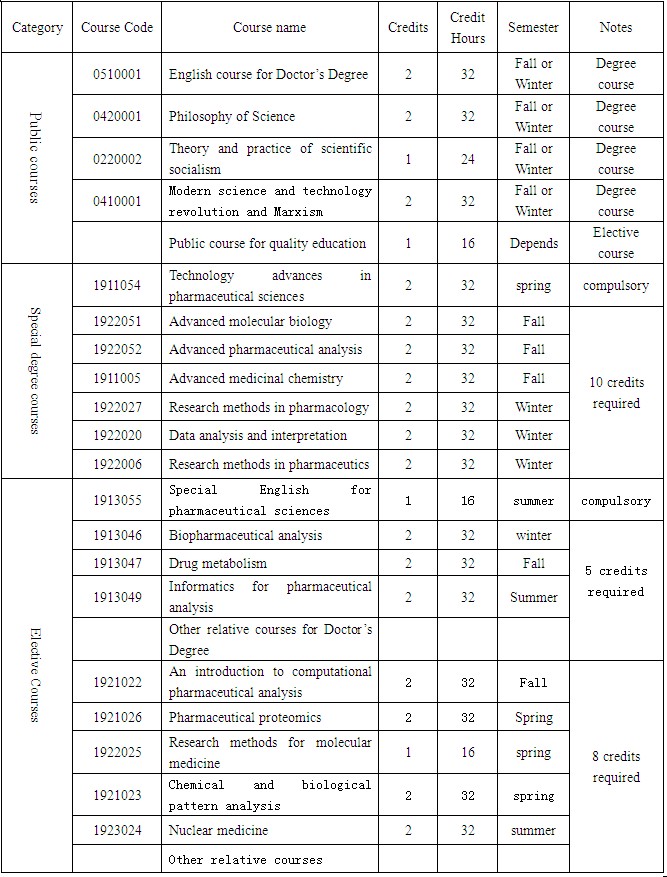
NEWS&EVENTS
-
09
2018.12
-
06
2016.12
-
02
2015.11
-
28
2015.10
-
07
2015.05
-
18
2014.12
