"UbitaADC": A Novel Approach to Enhance Drug Internalization in Cancer Cells
2026-03-30 | 药学院英文网
Antibody-drug conjugates (ADCs) are precision-targeted anticancer therapies comprising an antibody, a linker and a cytotoxic payload. By enabling the antibody to bind specifically to a target receptor, they deliver the cytotoxic payload directly into the tumour cells, thereby achieving a ‘precision strike’. However, the efficacy of ADCs depends not only on the binding affinity between the antibody and the target, but also heavily on the endocytosis and transport processes at the cellular level, particularly receptor-mediated endocytosis and subsequent delivery to the lysosome. In practical applications, many tumour-associated targets suffer from low efficiency of endocytosis and lysosomal delivery, which affects the efficiency of intracellular cytotoxin release and consequently limits the therapeutic efficacy of ADCs. Therefore, how to effectively enhance the endocytosis and lysosomal delivery efficiency of ADCs has become a major scientific challenge in this field.

On 25 March 2026, the research team led by Professor Liqiang Pan from the Laboratory of Precision Medicine and Biotechnology at the School of Pharmacy Zhejiang University, made significant progress in the field of ADC research. The findings were published in Cell Chemical Biology under the title “Receptor-ubiquitination-targeting antibody-drug conjugates for enhanced endocytosis and lysosomal delivery”. Addressing the challenge of effectively enhancing the endocytosis and lysosomal delivery efficiency of ADCs, the research team innovatively proposed a novel strategy known as “receptor-ubiquitination-targeting ADC” (ubitaADC). Through engineered design, this strategy enables the ADC molecule to recruit specific E3 ubiquitin ligases (RNF43/ZNRF3) whilst binding to target receptors on the surface of tumour cells, thereby inducing targeted ubiquitination of the receptor on the cell membrane. As one of the key intracellular protein regulatory signals, ubiquitination triggers the receptor to enter the endocytic pathway and directs its transport to the lysosomal degradation pathway. By leveraging this mechanism, ubitaADC transforms the traditional ADC process of ‘passive endocytic dependence’ into ‘active endocytic and lysosomal delivery’, thereby achieving precise control over the intracellular transport fate of the receptor.
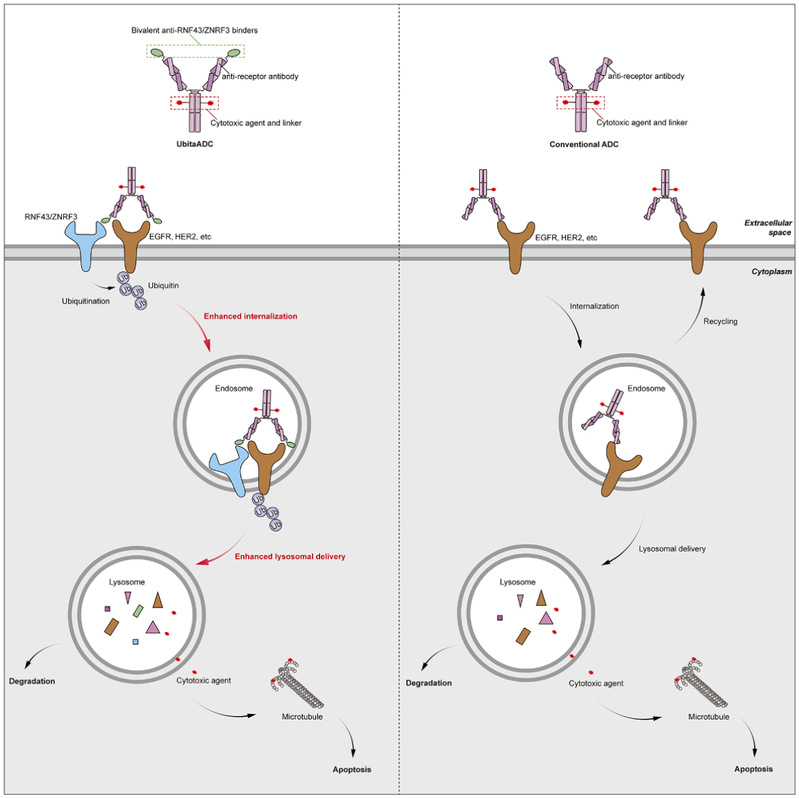
Using the epidermal growth factor receptor (EGFR) as a model target, the research team systematically validated the mechanism of action of ubitaADC. The results indicate that by recruiting E3 ubiquitin ligases, ubitaADC significantly enhances the level of EGFR ubiquitination, thereby accelerating its endocytosis and markedly improving its delivery efficiency to lysosomes. This process effectively promotes the release of the ADC drug within the cell, thereby significantly enhancing its cytotoxic effect on tumour cells. This study is the first to reveal, at the mechanistic level, the role of the ‘receptor ubiquitination–endocytosis–lysosomal delivery’ regulatory cascade in optimising ADC function. In in vivo anti-tumour experiments, ubitaADC also demonstrated superior therapeutic efficacy compared to conventional ADCs. Compared with the control group, ubitaADC was able to inhibit tumour growth more significantly, demonstrating stronger in vivo efficacy and potential clinical application value. Applying this strategy to another target, human epidermal growth factor receptor-2 (HER2), yielded similar results, further indicating that regulating receptor ubiquitination to reprogram intracellular transport pathways is a practical and universally applicable strategy for enhancing the therapeutic efficacy of ADCs.
Taking a novel approach, this study incorporates classical ubiquitination mechanisms into the design of antibody-drug conjugates (ADCs), proposing a new paradigm for ADC optimisation centred on the ‘regulation of receptor trafficking’. This strategy not only offers an effective solution to the problem of insufficient endocytosis efficiency in conventional ADCs, but also provides valuable insights for the design of targeted protein degradation agents and other types of conjugated drugs, such as nucleic acid conjugates and radiolabelled conjugates.
The School of Pharmacy Zhejiang University is the primary institution for this paper; Xinlei Zhuang, a postdoctoral researcher is the first author, and Professor Liqiang Pan is the corresponding author.
Original link:https://www.sciencedirect.com/science/article/pii/S245194562600070X?via%3Dihub
NEWS
-
20
2026.04
-
30
2026.03
-
10
2025.12
-
27
2025.11
-
25
2025.11
-
03
2025.11
