CPS-ZJU scientists propose a new strategy for protein modification on the surface of living cells
2023-11-06 | 药学院英文网
Autoimmune disease is a kind of inflammatory disease caused by abnormal immune state caused by the attack of normal cells by the body's own immune system, which eventually leads to tissue damage or organ dysfunction. Effective inhibition of immune inflammatory response is very important for the treatment of autoimmune diseases. Programmed cell death ligand 1 (PD-L1), which can inhibit T cell activity and alleviate immune response by binding to PD-1 receptors, has shown a wide range of applications in the treatment of autoimmune diseases in recent years. Therefore, increasing the engineering expression of PD-L1 on the surface of specific tissues / cells can transmit inhibitory signals, regulate the activity of tissue infiltrating T cells, prevent autoimmune attacks, and reshape the balance of the immune system. At present, the ways of engineering expression of specific proteins on cell surface mainly include genetic engineering, chemical reaction modification and physically acting membrane embedding and so on. Among them, the operation of expressing proteins on the cell surface with the help of genetic engineering is complex, and it is easy to be disturbed by the complex environment in the cells. With the help of cell membrane surface active functional groups such as amino and sulfhydryl groups to modify proteins on the cell surface by chemical reaction, it is easy to affect the cell activity, and the selectivity is poor. In recent years, the efficient modification of proteins on cell surface has been successfully realized based on unnatural sugar metabolism markers combined with biological orthogonal catalysis. However, due to the life activities such as cell metabolism and division, the retention time of proteins is limited, which further affects the further application. However, the modification of proteins on the cell surface is also faced with the problem of poor selectivity by the way of physical action membrane embedding. How to achieve long-term and specific modification of cell surface proteins is of great significance for the clinical application of related technologies.

In view of this, the team from CPS-ZJU, Jinhua Research Institute and the National key Laboratory of Advanced Drug Delivery System developed a dual-anchored cell surface engineering strategy, which combined with click chemistry and physical membrane insertion techniques to inhibit T cell activity by prolonging the retention time of immune checkpoint inhibitor protein PD-L1 on the surface of target cells such as islet β cells and chondrocytes. It has been proved that this strategy can effectively alleviate the incidence and symptoms of autoimmune diseases such as type I diabetes and rheumatoid arthritis in animal models, and the results were recently published in the journal Nature Communications. The first author of this paper is Shenqiang Wang, a postdoctoral fellow of CPS-ZJU, and the correspondent authors are Professor Jicheng Yu, Professor Jinqiang Wang and Professor Zhen Gu of Zhejiang University.
Firstly, the unnatural sugars containing azide groups (Ac4ManNAz) were delivered to the target tissue / cells by reactive oxygen species responsive nanoparticles, and the azide groups were modified on the cell surface by metabolic sugar engineering. Subsequently, dibenzocyclooctyne (DBCO) and PD-L1 co-modified with 1-distearoyl-sn--3-phosphatidylethanolamine (DSPE)-polyethylene glycol (PEG) were injected into the body, and the rapid and specific labeling of the target cells was realized by click chemistry. At the same time, the modified phospholipid long chain can also be embedded in the cell membrane by hydrophobic interaction, which overcomes the limitation that the existing techniques can not take into account both specificity and long-term effect. On the premise of achieving cell-specific labeling, the retention time of proteins on the cell membrane surface was significantly prolonged, thus achieving cell-specific and long-term labeling.
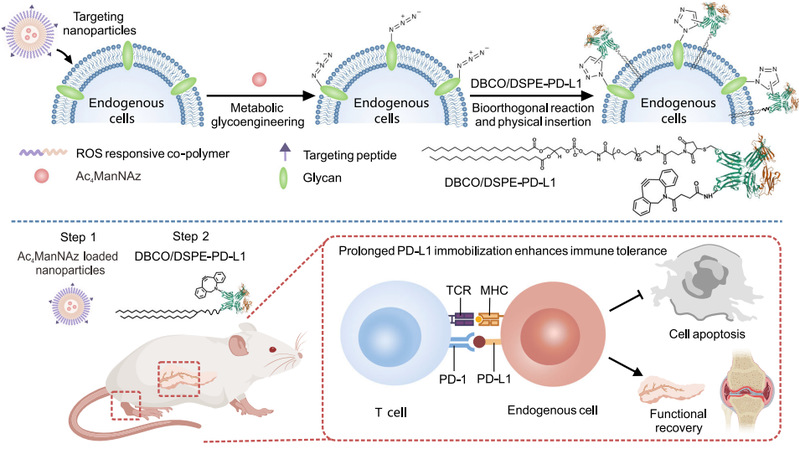
Figure 1.The dual-anchoring strategy prolongs the retention time of PD-L1 on the cell surface for the treatment of autoimmune diabetes and rheumatoid arthritis
This strategy can regulate immune homeostasis and enhance anti-inflammatory response by reducing leukocyte infiltration, inhibiting the expression of inflammatory factors and increasing local regulatory T cell population. The results of in vivo experiments showed that in the NOD model of non-obese diabetic mice, the incidence of diabetes could be significantly reduced by prolonging the modification time of PD-L1 on the surface of islet β-cells. In addition, in the model of rheumatoid arthritis, it is further verified that the treatment strategy can effectively alleviate the inflammatory response and reduce articular cartilage injury.
Original link: https://www.nature.com/articles/s41467-023-42725-1
Translator: Zesheng Cheng
NEWS
-
20
2026.04
-
30
2026.03
-
10
2025.12
-
27
2025.11
-
25
2025.11
-
03
2025.11
